
“I think he’s in the termite mound!” I shouted. Camila Calderón, a researcher at the Max Planck Institute of Animal Behavior, called back: “No! Let’s keep looking over this hill! Maybe the signal is bouncing!” Camila studies the greater spear-nosed bat, Phyllostomus hastatus (or just Phast to its friends). With a typical adult wingspan of a foot and a half, P. hastatus is one of the largest bats in the Neotropics. We were tracking a male that we had caught and equipped with a radio tracker the previous night. Now, we had followed the signal from that tracker into an overgrown valley where it appeared to come from a small termite mount about eight meters up a tree. Camila was not pleased.

P. hastatus typically live in harems, in which one male defends a roost with many females [1]. The typical number of females in a roost is around 18, but on rare occasions numbers as high as 100 have been reported [2]. Males not fortunate enough to live in a harem usually live in small bachelor groups. Camila’s goal was to put GPS trackers on all the females in a harem and study how information is socially transmitted amongst them. We were hoping this male would lead us to his harem, but there was no way a harem would fit in that termite mound. So, some wishful thinking: maybe the radio signal was just bouncing around the valley and we would find the harem over the next hill? This was not to be the case.
I circled around the tree with the termite nest and found a small opening at the bottom. I clicked on my high-beam red light and there was our bat, huddled up amongst several of a smaller bat species, Lophostoma silvicolum. The lack of a female colony was disappointing, but we were still witnessing an interesting behavior. P. hastatus roosts have been observed in caves, tree hollows, palm leaves, and human structures, but Barro Colorado Island, where we were working, is the only place individuals have been described living in termite mounds with L. silvicolum [2, 3]. Along with descriptions of male P. hastatus roosting alone or with other species in Brazil [2], this behavior suggests that the social organization of P. hastatus is far more flexible than once thought.

Most of our understanding of Phyllostomus hastatus comes from the extensive study of a few populations in Trinidad. The bats there roost in caves. Within a cave, many clusters of females can exist, but groups remain remarkably stable in composition; in an 18 month study of 61 females across four different harems, only five bats changed harems [1]. Even more fascinating, the females in these groups are not closely related. Young bats disperse from their parental groups within a year and appear to join or form new groups at random [1]. This means that kin selection (the benefit to the reproduction of your own genes when you help a relative) cannot explain any P. hastatus behavior that helps the group at a cost to the individual.

Yet, P. hastatus females don’t just live together, they forage together too! Individual P. hastatus tend to forage in the same general area night after night, and females from the same group forage adjacent to one another and frequently in each other’s turf [1]. The P. hastatus diet may have something to do with this. Though these bats are omnivorous, even eating other small mammals, they rely a lot on fruit and nectar when these resources are in season (and like my good friend the kinkajou, they have a long tongue for drinking nectar from flowers). These resources come and go quickly and may be hard to find, so searching in a group can be very advantageous. This is intuitive to us. We frequently split up when looking for things, but it comes at cost: you have to share whatever you find! Unless individuals are helping kin who share some of their genes, this only makes sense from an evolutionary perspective if individuals that share information about the location of food are more likely to receive similar information from their group-mates in the future. We call this process reciprocity, and it can lead to what looks like friendships between animals [4].

Though we do not yet have evidence that P. hastatus form reciprocal relationships with specific individuals, females that live in groups that forage together more have greater reproductive success [5]. We also know that our great spear-nosed friends can distinguish the calls of cave mates from bats in other caves, and calls of their own group from other groups in the same cave [6]. So, it seems likely that unrelated female P. hastatus forage cooperatively, reciprocally exchanging information about food availability. These bats are helping us understand how group living can evolve and be maintained even without the benefit of kin selection—this is super exciting! But we don’t yet know how information is disseminated through a group. Do individuals have preferred relationships and foraging partners? What kind of resources do they share information about? What kind of information do they share, and how do they do it?
That’s where Camila and her colleagues come in.
Recent research conducted by the lab of Camila’s adviser, Dr. Dina Dechmann, and Dr. Teague O’Mara in Bocas Del Toro, Panamá revealed a previously unknown foraging behavior of P. hastatus. Remarkably, the bats they tracked flew over twenty kilometers every day to forage at balsa trees (Ochroma pyramidale). Balsa trees produce an abundance of nectar-rich flowers from the months of December through February when food is often scarce [7]. The bats’ long-distance flights clearly indicate these flowers are an important resource for P. hastatus in Bocas Del Toro. Because group foraging is most beneficial when resources are highly concentrated and widely dispersed, like the balsa, this system has a lot of potential to reveal more about the social dynamics of group foraging. Camila and Dr. Dechmann are working together to learn whether group foraging occurs more when balsa flowers are in season, whether group foraging is reduced when balsa are more abundant and less patchy (like on Barro Colorado Island), and whether individual relationships are important for the reciprocal exchange of information.
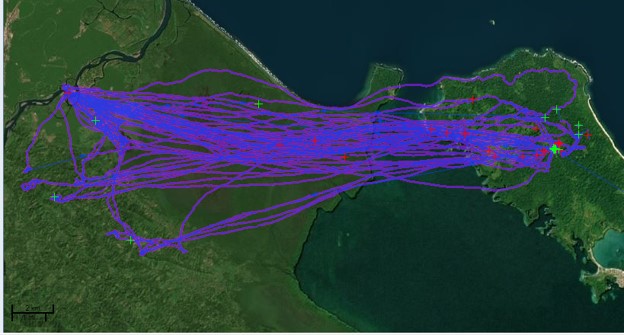
Even though Camila has not yet been able to track an entire group of females, bringing the study of greater spear-nosed bats to a new region is revealing how adaptable this species is. We have already seen that males differ in their dispersal patterns, sometimes roosting in the termite mound homes of other bats instead of cave-based bachelor groups. Does their foraging behavior change too? A lot more data is needed to know exactly what differences exist and why, but there is certainly a lot to discover!
Bats often get a bad rap—creatures of the night and all—but P. hastatus remind us: be kind to your neighbor. We all benefit when we help each other out.
Alexander Vining is a fourth year Ph.D. Candidate in the Animal Behavior Graduate Group. In affiliation with the Smithsonian Tropical Research Institute, he studies the spatial memory and movement of frugivorous mammals (including the kinkajou) on Barro Colorado Island, Panama. Alexander has a particular love for the elusive animals of the canopy and enjoys any research that brings him into the tree-tops. Follow his work on twitter @AVining_Opining.
All photos were contributed by Alexander Vining unless otherwise credited.
References
- McCracken, G. F. & Bradbury, J. W. (1981). Social organization and kinship in the polygynous bat Phyllostomus hastatus. Behavioral Ecology and Sociobiology, 8(1), 11–34. https://doi.org/10.1007/BF00302840
- Costa, L., Lourenço, E., Esbérard, C., & Silva, R. (2010). Colony size, sex ratio and cohabitation in roosts of Phyllostomus hastatus (Pallas) (Chiroptera: Phyllostomidae). Brazilian Journal of Biology, 70(4), 1047–1053. https://doi.org/10.1590/s1519-69842010000500019
- Kalko, E. K. V., Ueberschaer, K., & Dechmann, D. (2006). Roost structure, modification, and availability in the white-throated round-eared bat, Lophostoma silvicolum(Phyllostomidae) living in active termite nests. Biotropica, 38(3), 398–404. https://doi.org/10.1111/j.1744-7429.2006.00142.x
- Massen, J. J. M., Sterck, E. H. M., & De Vos, H. (2010). Close social associations in animals and humans: Functions and mechanisms of friendship. Behaviour, 147(11), 1379–1412. https://doi.org/10.1163/000579510X528224
- Boughman, J. W. (2006). Selection on social traits in greater spear-nosed bats, Phyllostomus hastatus. Behavioral Ecology and Sociobiology, 60(6), 766–777. https://doi.org/10.1007/s00265-006-0220-2
- Boughman, J. W., & Wilkinson, G. S. (1998). Greater spear-nosed bats discriminate group mates by vocalizations. Animal Behaviour, 55(6), 1717–1732. https://doi.org/10.1006/anbe.1997.0721
- Kays, R., Rodríguez, M. E., Valencia, L. M., Horan, R., Adam, R., & Ziegler, C. (2012). Animal Visitation and Pollination of Flowering Balsa Trees (Ochroma pyramidale) in Panama. 16(3), 66–68.